2025 Guide: Top 5 Equine Systemic Oral, Sensitivity-Guided Antibiotics in the USA — Evidence-Based Picks and How to Choose the Right One
Published on Monday, August 25, 2025
This category covers oral antibiotic options selected according to laboratory culture and sensitivity profiles to treat specific equine infections in the United States in 2025. Sensitivity-guided therapy prioritizes matching the drug to the identified pathogen and its susceptibility pattern, which improves clinical outcomes while supporting antimicrobial stewardship. Horse owners and veterinary professionals increasingly prefer sensitivity-guided oral choices because they reduce unnecessary broad-spectrum use, simplify dosing and administration for outpatient care, lower recurrence risk, and minimize drug residues important for performance and food-animal considerations. Practical factors that shape consumer preference in American markets include regional availability, veterinary prescribing practices, palatability and oral formulation (powder, suspension, tablets), withdrawal considerations, cost, and clear lab-to-clinic decision pathways.
Top Picks Summary
Why sensitivity-guided oral therapy matters
Laboratory-guided selection of oral antibiotics aligns treatment with the actual causative organism and its resistance profile, producing measurable benefits over empiric, broad-spectrum approaches. Research and stewardship guidance from veterinary and public health bodies emphasize targeted therapy to slow antimicrobial resistance, shorten infection duration, and reduce adverse effects. For equine practice, sensitivity guidance is especially valuable because many common pathogens show variable susceptibility across regions and patients, and oral formulations allow continued therapy outside the clinic while maintaining adherence.
Culture and sensitivity testing enables de-escalation from broad-spectrum to narrow-spectrum agents when appropriate, which is associated with reduced selection pressure for resistant organisms.
Studies and stewardship reviews show that targeted therapy, when started promptly after culture results, reduces treatment failure rates and recurrence compared with prolonged empiric therapy.
Optimized oral dosing schedules and palatable formulations improve owner compliance and complete-course adherence, factors linked to better clinical resolution and fewer relapses.
In USA, national antimicrobial stewardship initiatives and provincial veterinary guidelines increasingly recommend laboratory confirmation and sensitivity-guided treatment for systemic infections in horses.
Specific agents serve distinct roles: sulfonamide-trimethoprim combinations for broad coverage of common equine pathogens, metronidazole for anaerobic infections, tetracyclines/doxycycline for intracellular or atypical bacteria, and chloramphenicol reserved for particular resistant infections where legal and safety considerations permit use.
Frequently Asked Questions
Which equine systemic oral antibiotic fits sensitivity-guided use best?
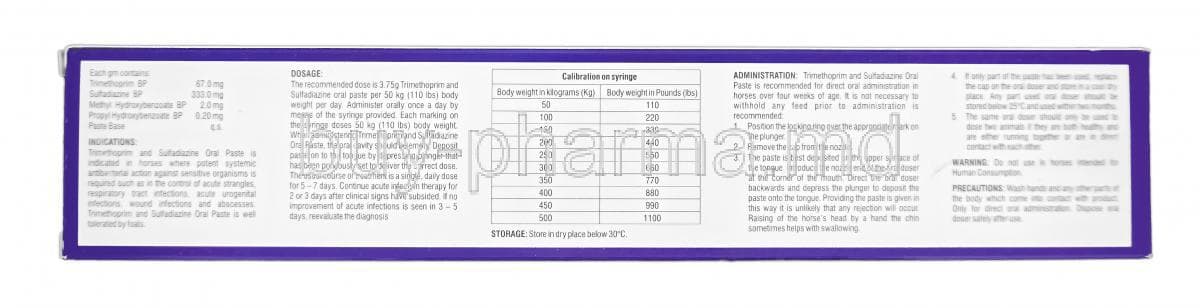
Trimidox Equine Oral Powder is the sensitivity-guided pick, combining trimethoprim-sulfadiazine broad-spectrum coverage with a powder formulation that mixes with feed for weight-based dosing for horses and groups.
What exact formulation does Trimidox Equine Oral Powder use?
Trimidox Equine Oral Powder is a powder formulation of a trimethoprim-sulfadiazine combination that can be mixed with feed, supporting reliable plasma levels when dosed by weight; it rates 4.2.
How do the oral options compare by price and rating?
Metronidazole 500mg Tablets list at $49.59 and have a 4.1 average rating; Trimidox Equine Oral Powder has a 4.2 rating, while Sulfadiazine Trimethoprim Oral Suspension has a 4 rating.
Which suspension option makes weight-based dosing easier after results?
Sulfadiazine Trimethoprim Oral Suspension uses an oral suspension form that eases precise weight-based dosing for variable-sized horses and supports targeted therapy guided by culture and sensitivity; it has a 4 average rating.
Conclusion
In the American 2025 context, a sensitivity-guided oral strategy gives horse owners and veterinarians the best chance of resolving systemic infections while supporting antimicrobial stewardship. The top options covered here — Trimidox Equine Oral Powder, Sulfadiazine Trimethoprim Oral Suspension, Metronidazole 500mg Tablets, Doxycycline Hyclate 100mg Tablets, and Chloramphenicol Palmitate Oral Suspension — each have defined roles: Trimidox for certain broad-spectrum indications and ease of dosing, Sulfadiazine Trimethoprim as a versatile first-line oral choice, Metronidazole for anaerobic disease, Doxycycline for intracellular or atypical pathogens, and Chloramphenicol Palmitate as a reserved option for specific resistant infections. For most sensitivity-confirmed, routine systemic equine infections in the USA, Sulfadiazine Trimethoprim Oral Suspension is often the best overall choice because of its broad applicability and established clinical performance when guided by lab results. We hope you found what you were looking for; use the site search to refine by infection type, drug formulation, provincial availability, or to expand into related topics like withdrawal times, compounding options, and stewardship resources.